The U.S. Food and Drug Administration has granted premarket approval for Allurion’s Gastric Balloon System with the swallowable Allurion Smart Capsule.
- The Allurion Smart Capsule is designed to be taken without surgery, endoscopy, or anesthesia.
- Once swallowed, the capsule is filled with fluid and remains in the stomach for roughly 4 months, helping patients feel fuller and reducing food intake.
- The company said the Capsule offers a safe and effective alternative to GLP-1 medications.
Allurion Technologies Inc. (ALUR) announced on Monday that the U.S. Food and Drug Administration (FDA) has granted premarket approval (PMA) for its Allurion Gastric Balloon System, clearing the way for the company to expand its non-surgical weight-loss treatment across the United States.

The FDA authorized the company’s PMA application for the system, which features the swallowable Allurion Smart Capsule.
How Does The Smart Capsule Work?
The Allurion Smart Capsule is designed to be taken without surgery, endoscopy, or anesthesia. Once swallowed, the capsule is filled with fluid and remains in the stomach for roughly 4 months, helping patients feel fuller and reducing food intake.
At the end of that period, a built-in release mechanism allows the device to deflate and pass naturally through the body. Patients may begin another treatment cycle about two months later. Unlike daily pills or weekly injections, the system provides several months of therapy from a single administration.
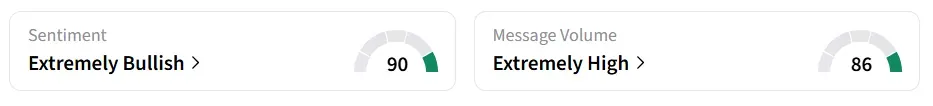
Following the update, Allurion’s stock traded over 50% higher on Monday morning. On Stocktwits, retail sentiment around the stock changed to ‘extremely bullish’ from ‘bullish’ territory the previous day. Message volume shifted to ‘extremely high’ from ‘normal’ levels in 24 hours.
Addressing A Large U.S. Market
More than 100 million Americans are affected by obesity, with approximately 80 million individuals falling within the body mass index range of 30 to 40 kg/m2, which qualifies them for the Smart Capsule.
“The Allurion Smart Capsule offers a safe and effective alternative to GLP-1s and bariatric surgery, and it should become a standard tool in the comprehensive treatment of obesity in the United States.”
-Shantanu Gaur, Founder and CEO, Allurion
The company also pointed to high discontinuation rates among GLP-1 medications, noting that many patients stop treatment within a year, often regaining lost weight.
ALUR stock has gained over 27% in the last 12 months.
For updates and corrections, email newsroom[at]stocktwits[dot]com.<
