The FDA has lifted a previous Complete Response Letter and resumed review of the company’s Biologics License Application for Deramiocel.
- The resubmission follows the positive results from the HOPE-3 Phase 3 trial.
- The FDA has set a Prescription Drug User Fee Act (PDUFA) target action date of Aug. 22, 2026.
- The investigational therapy aims to address both cardiac and skeletal complications of Duchenne muscular dystrophy.
Capricor Therapeutics (CAPR) announced on Tuesday a key development in its push to bring Deramiocel to market, a cell therapy for Duchenne muscular dystrophy (DMD) cardiomyopathy.

The U.S. Food and Drug Administration (FDA) has lifted a previous Complete Response Letter and resumed review of the company’s Biologics License Application (BLA), moving the therapy closer to potential approval.
FDA’s Review Rationale
The resubmission follows positive results from the HOPE-3 Phase 3 trial, in which Deramiocel met its primary endpoint and all Type I error-controlled secondary endpoints.
The FDA has classified the BLA as a Class 2 resubmission and set a Prescription Drug User Fee Act (PDUFA) target action date of Aug. 22, 2026. Capricor expects to be eligible for a Priority Review Voucher (PRV) if Deramiocel receives FDA approval.
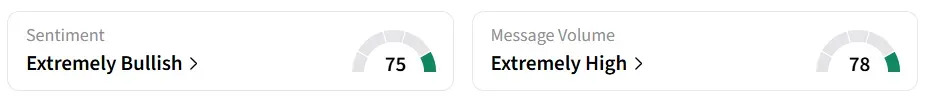
Capricor Therapeutics stock traded 17% higher on Tuesday morning. On Stocktwits, retail sentiment around the stock changed to ‘extremely bullish’ from ‘bullish’ territory the previous day. Message volume changed to ‘extremely high’ from ‘high’ levels in 24 hours.
Deramiocel’s Potential
The investigational therapy aims to address both cardiac and skeletal complications of DMD, potentially making it the first treatment to tackle the full spectrum of disease manifestations.
DMD is a serious genetic condition that mainly affects boys and causes muscles in the body to gradually weaken and break down.
“We believe the positive HOPE-3 results and broader clinical evidence reinforce Deramiocel’s potential to become a first-in-class therapy for Duchenne muscular dystrophy, with the opportunity to address both skeletal and cardiac manifestations of the disease.”
-Linda Marbán, CEO, Capricor Therapeutics
Deramiocel is made from heart-derived cells called cardiosphere-derived cells (CDCs). These cells help patients with DMDby reducing inflammation and preventing scarring by releasing tiny particles called exosomes that influence immune cells.
CAPR stock has gained over 189% in the last 12 months.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
