The CEO said Sellas is pursuing a “security force” immunotherapy strategy that trains immune cells to recognize cancer even as tumors evolve.
- CEO Angelos Stergiou said the company’s lead immunotherapy Galinpepimut-S (GPS), will attack leukemia cells regardless of genetic mutations.
- The therapy targets the Wilms Tumor-1 (WT1) antigen, thereby activating multiple T cells to eliminate leukemic stem cells and small residual cancer cells.
- Investors are closely watching the Phase 3 Regal trial, where final analysis will begin after 80 survival events.
Shares of Sellas Life Sciences Group Inc. (SLS) fell 1.5% in premarket trading on Monday as investors await a key readout from its late-stage leukemia study, while the company’s CEO said its GPS immunotherapy can attack cancer cells regardless of genetic mutations.

SLS stock jumped nearly 5% on Friday to hit $5.53 and added another 0.3% in extended trading.
Inside Sellas’ GPS Immunotherapy Approach
CEO Angelos Stergiou said in a LinkedIn post over the weekend that Sellas’ lead immunotherapy Galinpepimut-S (GPS) is designed to target leukemia regardless of genetic mutations.
Stergiou said GPS targets the Wilms Tumor-1 (WT1) antigen, a protein commonly expressed on leukemia cells, and is designed to activate multiple types of T cells, enabling the immune system to attack both leukemic stem cells and any remaining traces of cancer, regardless of the tumor’s genetic mutations.
“Targeted therapies are great, but they are inherently vulnerable to mutational evolution,” Stergiou said, adding that Sellas is developing a “security force” approach in which immunotherapy trains the immune system to recognize and eliminate cancer cells even as they change.
Stergiou also highlighted SLS009, a drug designed to block CDK9, a protein that helps cancer cells survive by supporting other growth-related proteins such as MCL-1 and MYC.
Phase 3 AML Trial Nears Final Analysis
Investor focus has centered on Sellas’ late-stage Regal trial, which is testing GPS as a maintenance treatment for patients with acute myeloid leukemia (AML), whose disease returned but later went back into remission after additional therapy.
The event-driven trial will assess whether GPS can improve overall survival. The final analysis will begin once 80 survival events are recorded. Sellas previously said the contract research organization monitoring the study had logged 72 events as of Dec. 26, bringing the trial closer to the threshold needed to trigger the final readout.
Maxim said that slower accumulation of events in survival-driven trials can sometimes reflect longer patient survival, although the study remains blinded until the data are formally analyzed.
How Did Stocktwits Users React?
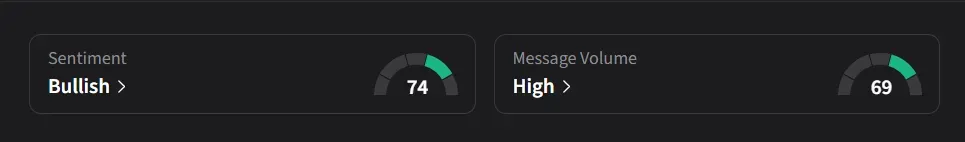
On Stocktwits, retail sentiment for SLS was ‘bullish’ amid a 68% jump in 24-hour message volume.
One user expects the stock to “drop today cause of how the market is going to be. Shorts will win cause of that.”
Another user said, “REGAL Phase 3 & SLS009 advancing — Two AML shots on goal — a lifeline for patients, hope for families, and upside for investors.”
SLS stock has surged over 360% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.<