The FDA has accepted Capricor’s resubmitted biologics license application, setting an Aug. 22 PDUFA decision date for Deramiocel in DMD.
- CEO Linda Marban dismissed concerns that Deramiocel would receive a conditional approval, citing a trial that met both primary and key secondary endpoints.
- The company is preparing for a potential launch, with its San Diego manufacturing capable of initially treating about 250 patients per year.
- Capricor reported a wider-than-expected Q4 loss of $0.62 per share and no revenue, but ended 2025 with about $318.1 million in cash.
Capricor Therapeutics, Inc. (CAPR) CEO Linda Marban has pushed back on regulatory doubts around its Duchenne Muscular Dystrophy (DMD) therapy Deramiocel, saying there is “no way” the drug ends up with only a conditional FDA approval, providing some

CAPR stock is heading for its best week since Feb. 20.
CEO Rejects Conditional Approval Risk For Deramiocel
On the company’s fourth-quarter (Q4) earnings call, CEO Linda Marban rejected the possibility that the therapy could receive only a conditional regulatory decision. “No. There’s no way this would be a conditional approval,” Marban said, noting that the trial supporting the application was a randomized, double-blind, placebo-controlled study that met its primary endpoint as well as key secondary endpoints.
The comments came after the U.S. Food and Drug Administration (FDA) accepted Capricor’s resubmitted biologics license application for Deramiocel, setting a PDUFA decision date of Aug. 22.
Capricor is seeking full approval of Deramiocel for Duchenne muscular dystrophy (DMD) and expects the data to support labeling that reflects benefits in both skeletal muscle and heart disease associated with the condition.
Launch Preparations Underway
The company is also preparing for commercialization if the therapy wins regulatory clearance. Capricor’s San Diego GMP manufacturing facility has already completed its FDA pre-license inspection and is positioned to support an initial commercial launch. The site currently has the capacity to treat about 250 patients per year.
Marban said the company plans to begin stockpiling commercial doses once labeling discussions with the FDA are finalized, signaling confidence in the potential approval.
Capricor is also expanding manufacturing capacity with additional clean rooms at the same facility, which the company expects could increase production to roughly 2,500 patients per year once fully operational.
Clinical Data Supporting Approval
Deramiocel’s application is supported by results from the Phase 3 Hope-3 trial, which met its primary endpoint measuring upper-limb function and key cardiac endpoints in patients with Duchenne muscular dystrophy. Additional data presented this week showed reduced cardiac fibrosis and improvements in functional measures tied to daily activities.
Marban said physician sentiment around the therapy has strengthened following the new data. “Physicians who before were hopeful are now convinced,” she said, adding that the company is receiving increasing inquiries about prescribing and availability.
CAPR Q4 Review
Capricor reported a Q4 loss of $0.62 per share, wider than analysts’ estimate of a $0.55 loss per share. The company reported nil revenue, compared with about $11.1 million a year ago, as milestone payments tied to its partnership with Nippon Shinyaku had already been fully recognized.
The company ended 2025 with about $318.1 million in cash, which will fund operations into late 2027.
How Did Stocktwits Users React?
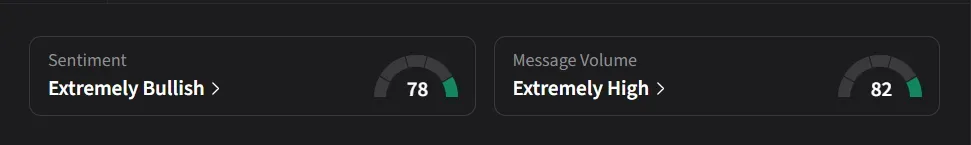
On Stocktwits, retail sentiment for CAPR was ‘extremely bullish’ amid over a 700% surge in message volumes over the past month.
One user said, “Congratulations to the Bulls, and to the patients who have been waiting for a miraculous discovery to treat their illness of DMD - Deramiocel, is just around the corner.”
Another user said interest in Deramiocel was strong, noting that participants are expected to join quickly and that others have been reaching out to try to take part, adding that “the demand is there for sure.”
The stock has surged over 150% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.<
